Thanks to biomedical engineer Mike Davis for writing an explanation of “Exosomes: what do we love so much about them?” for Circulation Research, a companion to his lab’s November 2016 publication analyzing exosomes secreted by human cardiac progenitor cells.
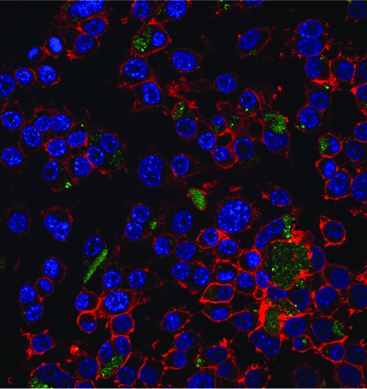
We can think of exosomes as tiny packages that cells send each other. They’re secreted bubbles containing proteins and regulatory RNAs. Thus, they may be a way to harvest the regenerative capacity of pediatric heart tissue without delivering the cells themselves.

Mike Davis, PhD is director of the Children’s Heart Research and Outcomes Center (HeRO), part of the Emory/Children’s/Georgia Tech Pediatric Research Alliance
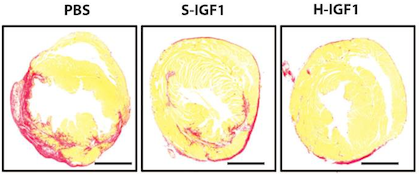
Davis’ lab studied cardiac tissue derived from children of different ages undergoing surgery for congenital heart defects. The scientists isolated exosomes from the cardiac progenitor cells, and tested their regenerative activity in rats with injured hearts.
They found that exosomes derived from older children’s cells were only reparative if they were subjected to hypoxic conditions (lack of oxygen), while exosomes from newborns’ cells improved rats’ cardiac function with or without hypoxia. Read more